|
8/17/2023 0 Comments Neon atomic structure
Therefore, for the Neon atom, the number of protons = atomic number = 10 The number of protons for any atom is always equal to the atomic number of that atom.Īs mentioned above, the atomic number of Neon is 10. 
Let us begin with calculating the number of protons. To draw the Bohr model we will first have to calculate the different atomic species present in the Neon atom. Now, using the above information we will draw the Bohr atomic model for the Neon atom. The electronic configuration of Neon is 2s 22p 6.The information that we can derive from the above-mentioned Neon box is as follows: Neon is a noble gas that belongs to the 18 th group of the periodic table: The Bohr model also states that the electrons have the capability to fall from higher to lower, as they release energy, or to jump from lower to higher energy levels, as they absorb energy.Īs we discuss the Bohr model of Neon, it contains 8 protons and 8 neutrons inside the nucleus, and 8 electrons revolving around the nucleus in 2 shells viz. The electrons present in this shell participate in the formation of bonds with other atoms. The electrons located in the shell farthest from the nucleus have the highest energy and are known as valence electrons while the shell is known as the valence shell. This is also the measure of the energy of the electrons that are said to be in the ground state when present in the smallest shell. 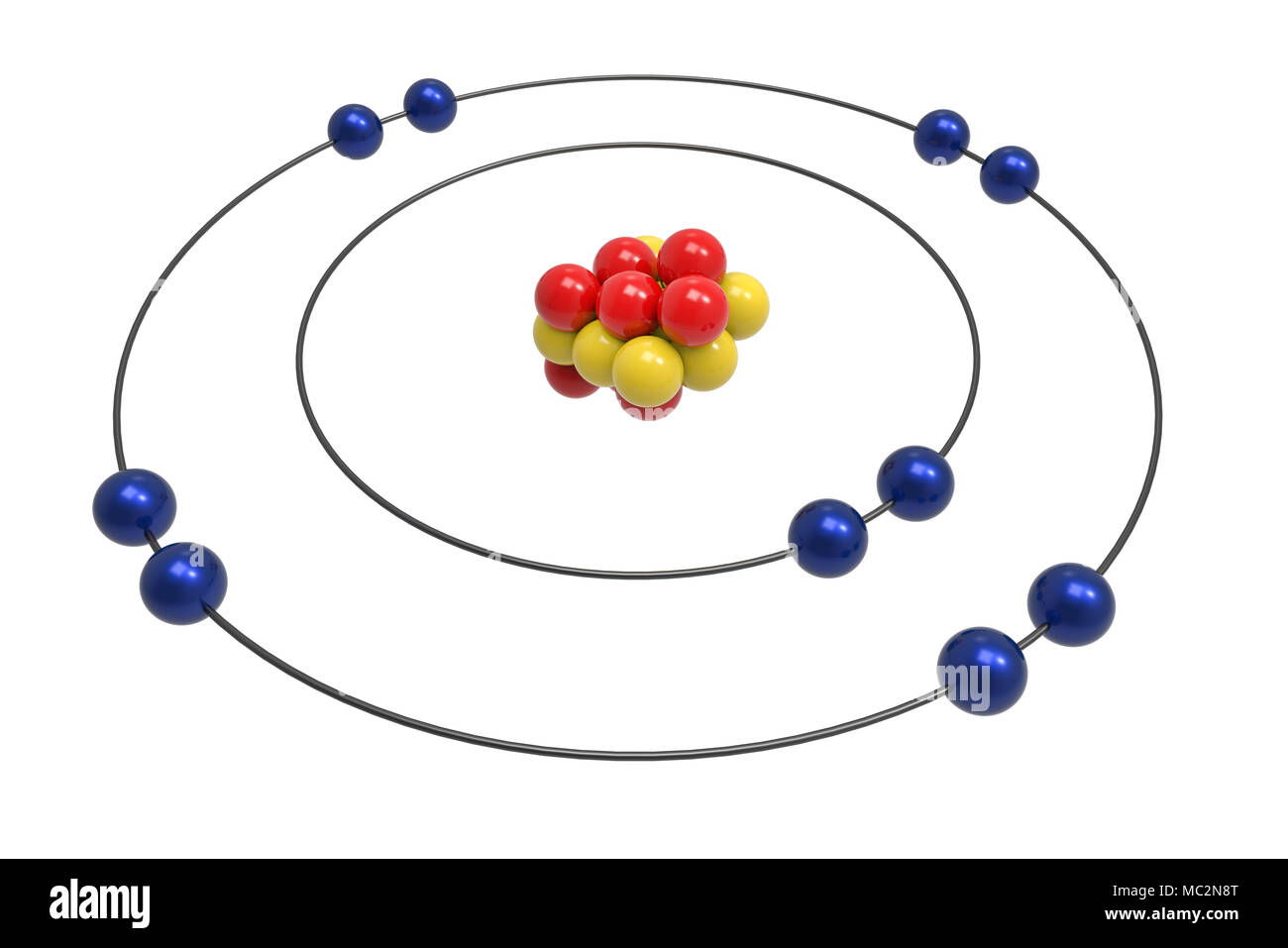
The lowest value is assigned to the shell located closest to the nucleus while the shell farthest from the nucleus has the highest value. The Bohr model names these shells as K, L, M, N, etc., or 1, 2, 3, 4, etc. Each electron has its specific energy level and each energy level has a specific number of electrons. These are also known as orbits or energy levels. 
Shells: The definite circular path followed by the electrons, inside an atom, around the nucleus.The electrons follow a definite path while revolving around the nucleus. Electrons: These are the negatively charged atomic particles that revolve around the nucleus.These are represented using the symbol n°. Neutrons: These are the charge-neutral particles located inside the nucleus.Protons: These are the positively charged species that are present in the nucleus.The nucleus in itself is a positively charged entity. Nucleus: It is located at the center of an atom and is constituted of two types of particles viz.The entire arrangement is exhibited in pictorial form for better understanding.īefore we start to explain the Bohr model of Neon, we must first understand a few terms related to the atomic structure. It also explained why electrons remain confined to their shells instead of falling inside the nucleus. This Bohr-Rutherford model explains the structure of the atom, placement of different atomic species inside the atom as well as the charge on different atomic particles. This model gave an explanation about the stability of electrons in their orbits and tried to overcome the drawbacks of the Rutherford model. This was termed as Bohr-Rutherford model. In 1913, the Rutherford model was again presented By Niel Bohr, however with certain important improvements. However, certain basic rules of classical mechanics and electromagnetic theory could not be answered using this model. This model depicted the atomic structure well, describing the location as well as the number of atomic species correctly. Rutherford’s model of the atom was proposed in 1911.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
